Snail baiting demonstration
The issue of snails in vineyards across multiple regions in WA is proving to be a persistent one.
Anecdotally it is believed that producers are not applying baits at the correct time (Autumn) mainly because of vintage respite, when snails are beginning to become active and laying eggs. Compounded by the wet and cool, spring has no doubt provided snail populations with ideal conditions to populate this year.
The purchase and application of baits is an expensive process; this activity funded by Wine Australia’s Regional Program (administered by Wines of WA) aims to improve producers understanding of the best methods of snail management through demonstration.
The activity consisted of two parts; a laboratory trial, and a field trial focusing solely on the brown garden snail (Cornu Aspersum).
Laboratory
A series of snail enclosures were set up with ten snails in each, replicated by three enclosures per treatment. Six (including a control) bait products were applied for each enclosure (see Table 1) and after 48 hours baits were removed and weighed.
| Trade name | Active ingredient |
| Control | Chicken pallet |
| Protect-Us™ | 10g/kg Iron Powder (organic product) |
| Metakill | 50 g/kg Metaldehyde |
| SlugOff® | 30 g/kg Metaldehyde (lentil shape pellet) |
| Slugout® | 18g/kg Metaldehyde |
| Baysol® | 20g/kg Methiocarb (registered for home gardens) |
Snails of various sizes were collected for this experiment from a bait and spray free vineyard and multiple home gardens.
The snails fasted on a moistened substrate of cotton bed sheet for 48 hours then placed into an enclosure on a fresh moistened substrate.
Snails were placed in the middle of the enclosure and baits were laid out around the snails to encourage feeding.
After 48 hours the baits were removed, dried and weighed to calculate quantity of bait consumed.
Counts of live and deceased snails were conducted at day seven by putting the snails in the middle of the container and the ones that didn’t move 24 hours later were counted as deceased.
Results and discussion
Data was analysed using analysis of variance (ANOVA) that incorporated the factorial treatment structure.
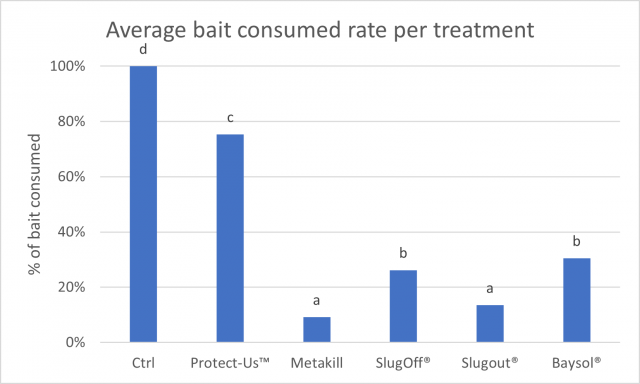
Figure 1 illustrates the percentage of bait consumed averaged across the three replicates. Bars with the same letter do not differ significantly (P<0.05) according to Fisher’s least significant difference test.
The graph clearly shows there is significantly less bait consumed compared to the control (P<.001).
Of the baits, Protect-Us™ was consumed much more than the other four baits.
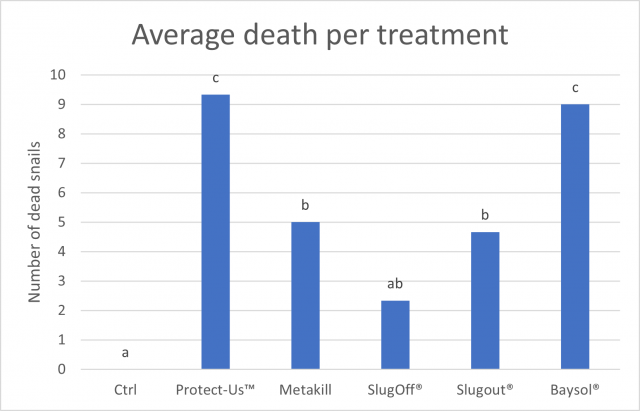
As Figure 2 shows, all baits were effective in killing snails when compared to the control (P<0.001). All baits except for SlugOff® caused significantly more snails to die than the control. Protect-Us™ caused the highest death rate, followed by Baysol®. Different concentrations within the metaldehyde based baits did not cause significant differences in mortality rates when compared to each other.
No strong relationship between the amount of bait consumed and mortality rate was observed.
Field
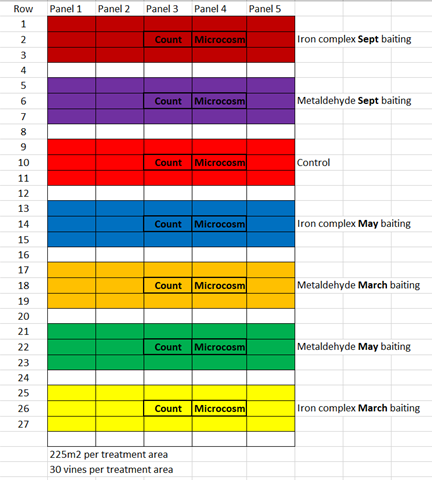
The field component of this activity was set at a commercial vineyard in the Margaret River wine region. A one hectare block of Sauvignon Blanc was utilised to demonstrate the effectiveness of two bait products; an iron complex based bait representing an organic product and a metaldehyde based bait representing a conventional product. Three separately designated treatment areas were marked and baits applied at three different timings; March, May and September (as shown below).
These baits were manually applied (at label rate), Metakill was applied at 10 kg per hectare and Protectus™ at 15kg per hectare.
The middle panel for each treatment zone was monitored for snail numbers in both the canopy and vineyard floor. Microcosms were also installed into each treatment zone with 20 snails enclosured so to observe mortality rate of a contained population within the zone.
Snail numbers (both live and dead) were observed in the monitoring panels as well as in the microcosms before application, 24 hours later and 7 days after bait application.
This vineyard was also used to collect monthly samples for dissection to monitor life cycle stages that may indicate timing of egg laying (via albumen gland size). The snail management demonstration commenced in March 2021 and will be completed in February 2022.
Results and discussion
The results from this field component are for demonstration purposes only as there is not sufficient replications for the data to be statistically validated.
There is a trend that the number of snails in the canopy gradually increased from March to September.
During counting it was observed that there were no small snails (shell width less than 10mm) in March, however many snails of this size were observed in May and September. There were also more medium sized snails (between 10mm-15mm) from May to September.
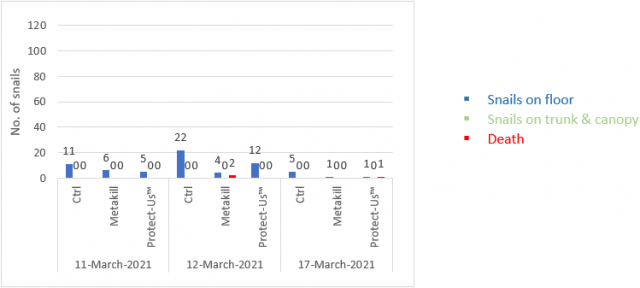
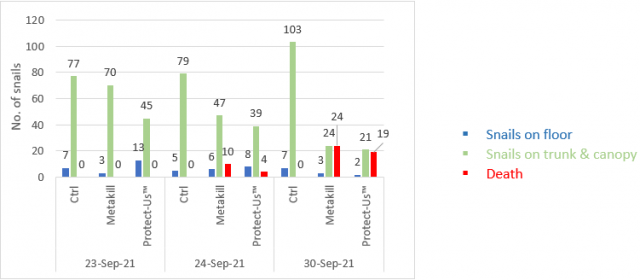
The following figures illustrate the observations made in March (Figure 4), May (Figure 5) and September (Figure 6) of snail populations within the monitored panels.
No snails were observed in the canopy in March, this is attributed to the block being harvested days before the count. There were also relatively low numbers found on the vineyard floor and few deaths.
May populations increased but very few signs of snail death after the baits were applied.
September observed an influx of snails in the canopy and on trunks and after seven days, both baited treatments showed a decline in snails on the vines and almost equal proportions of dead snails to those remaining on the vines.
Observations from the populations enclosed in the microsomes are summarised in Figure 7. The timing of these counts mirrored those made in the monitored panels.
An anomaly should be noted in regard to the May data set being that sheep within the block at the time did disturb the microcosms and potentially allowed some of the enclosed snails to escape.
The baits appear to have similar efficacy but in terms of ideal bait timing, it is difficult to comment based on variables such as vegetation within the microcosms providing an alternative food source and weather events influencing activity.
Life cycle observations from this activity will be reported in a future newsletter edition.
For more information on this work, contact Yu-Yi Liao, Technical Officer or Richard Fennessy, Research Scientist.
Acknowledgements
This activity is funded via the Wine Australia Regional Program which is administered by Wines of WA. Support from Mike Sleegers at Cowaramup Agencies and AHA Viticulture in providing access to a site and bait products is also greatly appreciated.